Round 2 -Meta-Analysis Internship
A Practical and Interactive Course on Meta-Analaysis
Systematic reviews and meta-analyses are research methods that combine and analyze data from multiple studies conducted on similar research topics. These methods have been widely used in various fields, including cancer, infectious disease, and social sciences, to name a few. They are powerful tools that can substitute the difficulties in performing large-scale randomized controlled trials. However, it is important to note that the inclusion of studies with any biases or improperly assessed quality of evidence in systematic reviews and meta-analyses could yield misleading results. Meta-analysis, which is a mathematical tool that allows researchers to mathematically combine outcomes from multiple studies, has been ranked among the top research methods in terms of quality and validity.
analyze data from multiple studies conducted on similar research topics. These methods have been widely used in various fields, including cancer, infectious disease, and social sciences, to name a few. They are powerful tools that can substitute the difficulties in performing large-scale randomized controlled trials. However, it is important to note that the inclusion of studies with any biases or improperly assessed quality of evidence in systematic reviews and meta-analyses could yield misleading results. Meta-analysis, which is a mathematical tool that allows researchers to mathematically combine outcomes from multiple studies, has been ranked among the top research methods in terms of quality and validity.
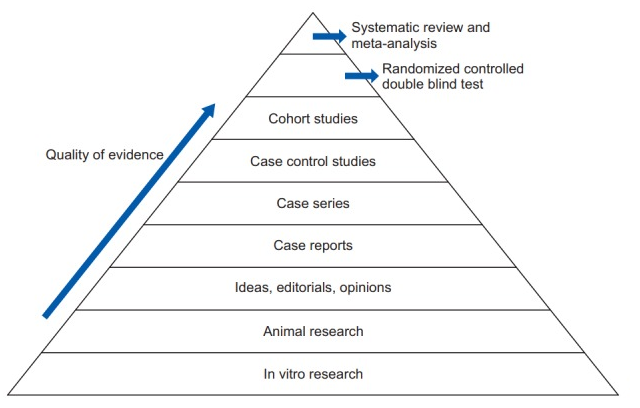
It is easy to confuse systematic reviews and meta-analyses. A systematic review is an objective, reproducible method to find answers to a certain research question, by collecting all available studies related to that question and reviewing and analyzing their results. A meta-analysis differs from a systematic review in that it uses statistical methods on estimates from two or more different studies to form a pooled estimate. Following a systematic review, if it is not possible to create a pooled estimate, it can be published as is without progressing to a meta-analysis; however, if it is possible to create a pooled estimate from the extracted data, a meta-analysis can be attempted. Systematic reviews and meta-analyses usually proceed according to the flowchart:
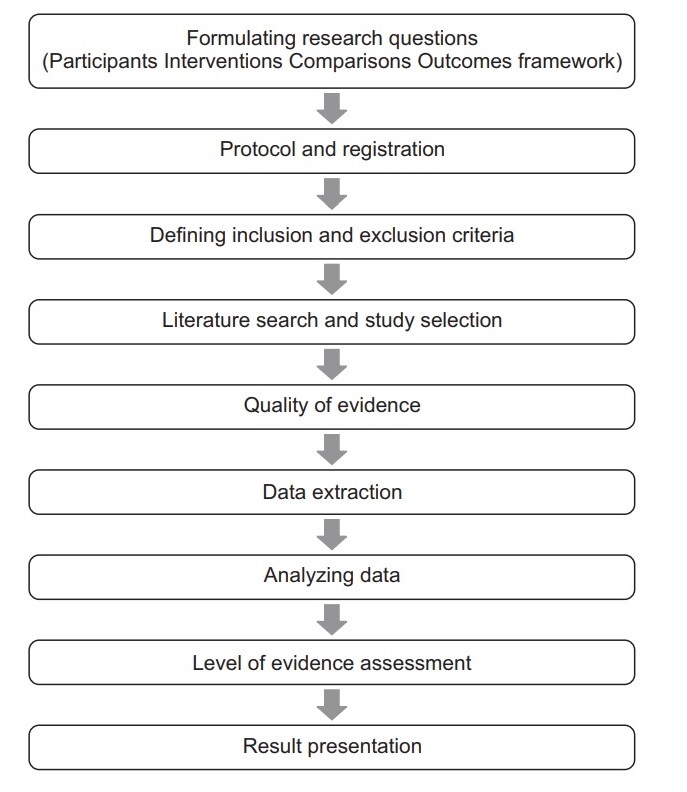
The aim of this internship program is to help the prospective intern go through all the steps of systematic review with the help of our dedicated research assistants and lecturer. The program takes three to 5 months. Each intern will choose a topic, the topic is evaluated for novelty by a peer in the field of the topic. If the topic is approved, a research assistant will be assigned to the intern to help him/her go through all the steps, including preparing the final report and the scientific article ready for submission to the proper journal.
This course is designed for health services researchers, epidemiologists, statisticians, systematic reviewers and decision analysts who are planning to perform a meta-analysis of their own concern.
The systematic review and met-analysis internship program is design to help the prospect intern to go through the all steps of choosing a proper topic, running a systematic review and meta-analysis and publishing the result. The program is divided into two main modules 1) Teaching and lecture-based modules, and 2) interactive and assistant-based modules. The lecture-based modules include 18 hours of lectures covering the principles of systematic review, and meta-analysis and the interactive based modules include the main part of the course in which an experienced research assistant is assigned to intern to help him through the process of selecting a real-world topic, do the search, evidence analysis, statistical analysis and ultimately reporting and preparing the final product “a manuscript ready for submission to a proper journal.
A basic understanding of common statistical concepts (e.g., confidence intervals and hypothesis tests) and, though not essential, some prior knowledge of SR and meta-analytic concepts.
The whole program is divided into following components:
- Teaching Module
- Topic selection (Interactive and assistant based)
- Performing systematic review and meta-analysis with help of experience research assistant ((Interactive and assistant based)
- Writing report and preparation of manuscript for publication with the help of experience research assistant (interactive and assistant based).
The Teaching Modules:
Principles of systematic review and evidence analysis:
The focus of this course is on advanced topics in evidence synthesis, including state-of-the-art methods such as GRADE. The workshop consists of a mixture of interactive presentations, small-group discussions, and hands-on computer exercises.
The course aims to introduce participants to the methodology of systematic review (SR). By the end of the course participants should be able to:
- Explain the need for systematic review
- Distinguish between and mention the advantages and disadvantages of the various types of literature review
- Formulate a systematic review question
- Appreciate the role of tools to assess risk of bias (RoB), including their application to RCT
- Summarize the findings of a systematic review;
Assess the quality of evidence in a SR using GRADE approach;
This module includes six lectures as follows:
- Lecture 1: Explain the need for systematic review;
- lecture 2: Distinguish between the advantages and disadvantages of the various types of literature review;
- Lecture 3: Formulate a systematic review question;
- Lecture 4: Appreciate the role of tools to assess risk of bias (RoB), including their application to RCT;
- Lecture 5: Summarize the findings of a systematic review;
- Lecture 6: Assess the quality of evidence in a systematic review using GRADE approach
Recommended teaching materials:
- Egger M, Davey Smith G, and Altman D. Systematic Reviews in Health Care: Meta-analysis in Context, 2nd Edition, BMJ Books, 2001.
- Higgins JPT, Green S (editors). Cochrane Handbook for Systematic Reviews of Interventions Version 5.1.0 [updated March 2011]. The Cochrane Collaboration, 2011.
- GRADE working group. GRADE Series. Journal of Clinical Epidemiology. Available at: http://www.jclinepi.com/content/jce-GRADE-Series
The Principals of meta analysis
This module includes three parts
Part I- Meta-analysis – Meta Analysis Basic
This part includes the following lectures’ topic:
- Basic concepts of meta-analysis
- Effect measures, commonly used in meta-analysis of intervention studies
- Modeling strategies for meta-analysis: fixed-effect models (I-V, M-H, Peto) vs. random-effects model [D-L, P-L, H-K-S-J]
- Assessment of heterogeneity
Suggested Readings:
- White IR, Schmid CH, and Stijnen T, Choice of effect measure and issues in extracting outcome data, in Handbook of Meta-Analysis, C.H. Schmid, T. Stijnen, and I.R. White, Editors. 2020, Chapman and Hall/CRC Press: USA. p. 27-38.
- Röver C, Knapp G, and Friede T. Hartung-Knapp-Sidik-Jonkman approach and its modification for random-effects meta-analysis with few studies. BMC Med Res Methodol, 2015; 15: 99. DOI: 10.1186/s12874-015-0091-1.
- Harris RJ, Deeks JJ, Altman DG, et al. Metan: Fixed- and Random-Effects Meta-Analysis. Stata J, 2008; 8(1): 3-28. DOI: 10.1177/1536867×0800800102.
- Thompson SG and Sharp SJ. Explaining heterogeneity in meta-analysis: a comparison of methods. Stat Med, 1999; 18(20): 2693-708.
Part II- Meta-analysis (Subgroup analysis)
This part includes the following lectures topic
- Subgroup analysis vs. sensitivity analysis and credibility of subgroup effects (ICEMAN tool)
- Meta-regression and aggregation bias in meta-analysis
- Publication bias and small study effect (statistical tests and visual inspection with funnel and contour-enhanced funnel plots)
- Meta-analysis of prevalence and proportions
Suggested Readings:
- Schandelmaier S, Briel M, Varadhan R, et al. Development of the Instrument to assess the Credibility of Effect Modification Analyses (ICEMAN) in randomized controlled trials and meta-analyses. CMAJ, 2020; 10; 192(32): E901-E906.
- Baker WL, White CM, Cappelleri JC, et al. Understanding heterogeneity in meta-analysis: the role of meta-regression. Int J Clin Pract, 2009; 63(10): 1426-34. DOI: 10.1111/j.1742-1241.2009.02168.x.
- Thompson SG and Higgins JP. How should meta-regression analyses be undertaken and interpreted? Stat Med, 2002; 21(11): 1559-73. DOI: 10.1002/sim.1187.
- Peters JL, Sutton AJ, Jones DR, et al. Contour-enhanced meta-analysis funnel plots help distinguish publication bias from other causes of asymmetry. J Clin Epidemiol, 2008; 61(10): 991-6. DOI: 10.1016/j.jclinepi.2007.11.010.
- Peters JL, Sutton AJ, Jones DR, et al. Performance of the trim and fill method in the presence of publication bias and between-study heterogeneity. Stat Med, 2007; 26(25): 4544-62. DOI: 10.1002/sim.2889.
- Nyaga VN, Arbyn M, and Aerts M. Metaprop: a Stata command to perform meta-analysis of binomial data. Arch Public Health, 2014; 72(1): 39. DOI: 10.1186/2049-3258-72-39.
Part III- Meta-analysis – Network Meta-analysis (NMA)
This part includes the following lectures
- Understand what indirect comparisons and NMA are and why they are used;
- Perform NMA using Stata, with continuous and dichotomous data;
- Understand the assumptions made in NMA and use Stata to examine consistency;
- Be aware of different techniques to present the results from NMA;
- Be able to critically appraise a paper that uses NMA;
- Evaluate the quality of evidence in a NMA.
Reporting and writing manuscript of systematic review and meta-analysis:
This part of the internship is lecture based, interns will develop the skills on how to write a scientific paper and how to report the result if systematic review and meta-analysis. The part includes the following lecture’ topics:
- Writing and Publication Process
- Basic Writing Principles
- Abstract
- Introduction Section
- Methods Section
- Results Section
- Discussion Section
Each intern is supposed to choose his own topic of systematic review and meta-analysis. Interns may share a topic with each other and agree on the share authorship. This will help the intern to have more product and publication at the end of its internship.
In order to have a successful outcome, the topic needs to be a novel topic and carry a substantial degree of novelty. The topic will be scrutinized by our faculty and the interns need to defend his topic and make sure the novelty the topic, the needs of systematic review and meta-analysis is out there, etc. There will be one lecture on how to choose a topic of scientific worthy for systematic review or meta-analysis. We encourage intern to spend time in choosing a proper topic.
Performing a real-world systematic review and meta-analysis (Interactive with help of experience research assistant).
This part of the internship is, in fact, the main part, the intern who has chosen his topic will b have a research assistant assign to him/her. The research assistant will go through the whole process of systematic review (right database, search strategies, evidence analysis, statistical analysis). Our research assistants are experts in meta-analysis, and they have published and contributed to many systematic reviews.
Writing report and preparation of manuscript for publication with the help of experience research assistant.
This part of the internship complements the entire process of lectures, search, evidence analysis, statistical analysis. The same research assistant, who has been with the intern during the course, will help the intern to prepare his report and write a proper manuscript for publication.
| The first round of the program included 10 Interns, which resulted in 14 publications. |
The Faculties:
Alireza Mosavi Jarrahi, MSPH, Ph.D.
Prof. of Epidemiology
Dept of Social Medicine, Medical School, Shahid Beheshti University of Medical Sciences, Tehran, Iran
Past President-elect, Asian Pacific Organization for Cancer Prevention,
Chief Executive Officer, West Asia Organization for Cancer Prevention- APOCP’s West Asia Chapter
Seyed Saeed Hashemi Nazari, MD, Ph.D.
Professor of Epidemiology
Department of Epidemiology, School of Public Health and Safety
Prevention of Cardiovascular Disease Research Center, Imam Hossein Hospital
Shahid Beheshti University of Medical Sciences
Prof. Amina Mohammed Al Marzouqi, Ph. D.
Prof. of Health Policy,
Vice Chancellor at University of Sharjah
College of Health Sciences, University of Sharjah, Sharjah, UAE
Syed Aziz Rahman, Ph.D
Associate Professor of Health Managment
College of Health Sciences, University of Sharjah, Sharjah, UAE
Clinical Assistant Professor, University of British Columbia, Canada
Nabeel Al-Yateem, RN, Ph.D.
Associate Professor of Nursing
College of Health Sciences,
University of Sharjah,
Sharjah, UAE
Dr.Ahmed Hossain, MS, Ph.D.
Associate Professor of Biostatistics and Epidemiology
College of Health Sciences,
University of Sharjah,
Sharjah, UAE
Mitra Zandi, BScN, MScN, Ph.D
Associate Professor of Medical Surgical Nursing
Head of International Affairs of the Nursing & Midwifery Faculty
Shahid Beheshti University of Medical Sciences, Tehran, Iran
Zalikha Khamis Darwish Al-Marzouqi, MS, Ph.D.
Senior Science Teacher
Oman College Of Health Sciences
North Batinah Branch, Oman
The Systematic review and meta-analysis internship program is organized by the West Asia Organization for Cancer Prevention, APIOCP’s west Asia Chapter. The program is implemented by very experienced research assistants and lecturers in the field of evidence based analysis from different universities. The program best fit to health services researchers, epidemiologists, statisticians, systematic reviewers and decision analysts who are planning to perform a systematic review or meta-analysis of their own topics.
The prospect intern needs the basic understanding of common statistical concepts (e.g., sampling, variations, confidence intervals and hypothesis testing) and, though not essential, some prior knowledge of systematic review and meta-analytic concepts. Normally, young and early career scientists are the main beneficiaries of the internship program however, graduate student of different discipline can participate.
The program is in English and the prospect intern needs to have English writing and listening skills to participate in the lectures and teaching assistant sessions.
To participate, you need to register and send your resume and a statement of purpose. The statement of purpose would describe the interns aim and goal as well as its capability and better include a topic for undertaking a systematic review or met-analysis.
The program may take up to 6 months (though the course organizer tries to help the intern to finish the program in 4 months).
Each intern will be assigned an experienced Research Assistant. The RA will couch and help the interns in all steps of the systematic review and meta analysis, PLUS preparing the manuscript, submitting the manuscript to the proper journal and making sure it will be published. The close partnership and engagement of RA with the intern throughout the course of the program is, in fact, the part that make this internship different from other similar workshop or educational activities).
While the lectures are set at a fixed time (the intern must be ready to attend), the interactive part is flexible to arrange by the intern and his/her teaching assistant.
Each round of the program take 25 Interns.
The registration fee for the whole internship program, depending on country of residency, is 200 to 600 US dollars per intern. The UN economic categories of income is the based for the fee (Low income = 200, Middle income = 400 and high income = 600)
There are three scholarships that funds three interns registration fee. The scholarship is provided by the College of Health Sciences, University of Sharjah, Sharjah, UAE. Registration is open and will be closed on Jan. 10th, 2024.
| Registration is now open. To register, just click on “Registration Form” Tab. |
The registration is closed now.